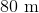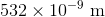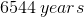All College Physics Resources
Example Questions
Example Question #1 : Quantum And Nuclear Physics
A car with mass 

The de Broglie wavelength is given by 
Example Question #1 : Radioactive Decay
Radiocarbon dating is a method that allows scientists to estimate the age of organisms after they have died. A fairly constant amount of radioactive 




If a scientist finds that a fossil contains 

In this question, we're given a brief description of radiocarbon dating. We're given the amount of 
First, let's briefly go over radiocarbon dating. This method essentially assumes that the amount of radioactive carbon within an organism remains fairly stable at any given time while the organism is alive. Moreover, this amount of radioactive carbon is related to the amount of radioactive carbon in the atmosphere. Once the organism dies, however, it ceases to gain any radioactive carbon; rather, the 


Since we're dealing with the decay of 
We can further rearrange this expression to isolate the variable for time.
In arriving at this expression, we see that we need to know the rate constant, 

Rearranging, we can find the rate-constant.
Now that we have the rate constant, we can plug this value into the previous expression to solve for 
Example Question #2 : Radioactive Decay
Suppose that 



Note: The half-life of the drug is 
For this problem, the first step is to find the rate constant for the decay reaction. Since we're given the half-life, we can calculate this value using the following equation.
Now, we can use the first order rate equation to find out how much drug will be left after the first time interval of 
So, after the first 



With this new amount in mind, we'll need to calculate how much of the drug will be present after more time has elapsed. We can do this by using the same equation as before.
This is the final amount of drug that is expected to be present in the patient's body at that instant of time.
Example Question #1 : Radioactive Decay
Iodine-131 has a half life of 8.02 days and undergoes beta decay.
If a 






Finding the amount is a simple half life problem. Start with setting up what you know for any half-life problem.


From there simplify.

Now plug this into the equation and find what happens after 7 days.
Beta decay is when a neutron releases an electron and becomes a proton. Therefore, the atomic number goes up by one but the atomic mass remains the same. This results in xenon-131.
Example Question #1 : Mechanics
A car traveling at 


The mass of the car is required
Kinetic energy is given by


Next, we will calculate the final energy of the car, also in terms of the unknown mass of the car:

To find the ratio of the final to initial kinetic energy, we divide 






Example Question #1 : Work And Energy
A 


So in order to solve this problem, we first should think about where all the energy in the system is coming from and going. Specifically when you pull the spring back, the work you do is turned into potential energy stored in the spring. So:
and the energy transforms into kinetic when you let go, but some of that energy is lost to friction so

So use the equation below, enter in your known quantities and then solve for the work done by friction.
Plug in known values and solve.
Example Question #2 : Work And Energy
A toy car is set up on a frictionless track containing a downward sloping ramp and a vertically oriented loop. Assume the ramp is 
What additional piece of information is necessary to calculate the maximum height of the loop if the car is to complete the loop and continue out the other side?
None
The value of
The mass of the car
The distance between the end of the ramp and entrance to the loop
The exact shape of the loop
None
This is an example of conservation of energy. The car starts at the top of the ramp, at height 



At the bottom of the loop, all of the potential energy will have been converted into kinetic energy.
As the car traverses the loop and rises above the ground, kinetic energy will be converted back into potential energy. The shape of the loop does not matter in this case -- only the vertical distance between the ground and the car.
In the tallest possible loop, all kinetic energy at the bottom is converted to potential energy at the top. This is the maximum height the car can reach -- there is no additional energy left to continue climbing a taller loop. Therefore, the potential energy at the top of the tallest loop we can build is equal to the kinetic energy at the bottom of the loop. But we have already noted that the kinetic energy at the bottom of the loop is equal to the potential energy at the top of the ramp.
Therefore, we set 






Example Question #1 : Mechanics
An elevator is designed to hold 


Power is the rate of energy transfer. To raise a 




Example Question #2 : Conservation Of Momentum And Energy
How far can a person jump while running at 

We know that:
and we are looking for the maximum height (vertical displacement) this person can obtain, so we aren't concerned with 
We can apply the conservation of energy:
Masses cancel, so
Solve for 

so let's plug in what we know

Example Question #3 : Conservation Of Momentum And Energy
If a 


Let's first write down the information we are given:
In order to solve this problem we must apply the conservation of energy, which states 
This means that as the project reaches its max height energy is converted from Kinetic Energy (energy of motion) to potential gravitational energy (based off of height).
We can subtract 



so we can solve for the height
where
therefore
All College Physics Resources