All GRE Subject Test: Chemistry Resources
Example Questions
Example Question #211 : Gre Subject Test: Chemistry
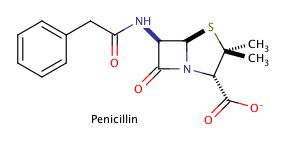
Shown above is the chemical structure for penicillin, a common prescription drug. How many chiral carbons does penicillin have?
Two
Zero
Five
One
Three
Three
The correct answer is three. The key to finding chiral carbons is to look for carbons that are attached to four different substituents. We can immediately eliminate any carbons that are involved in double bonds, or that have two hydrogens attached. Given this, we find that there are three chiral carbons. Note that carbon chains of varying content will qualify as different substituents, allowing chiral carbons to bond to two other carbons.
Example Question #2 : Stereochemistry
Compounds that are mirror images of each other are called __________.
conformers
enantiomers
stereoisomers
diastereomers
enantiomers
Stereoisomers are isomers that differ in the orientation of atoms in space, but have the same bonding patterns and structures. Enantiomers are a specific class of stereoisomers that differ in orientation around a chiral center to create mirror image molecules.
Diastereomers are a type of stereoisomer that are not related through a reflection operation, and may differ at more than one chiral center. Conformers have the same structural formula, but different shapes due to bond rotation.
Example Question #221 : Gre Subject Test: Chemistry

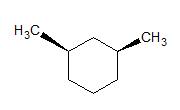
The given molecules are __________.
stereoisomers
constitutional isomers
None of these
identical
conformers
stereoisomers
Stereoisomers have different orientations around a single stereocenter. The two molecules are stereoisomers. Specifically, these molecules are epimers, meaning that they differ at only one stereocenter.
Constitutional isomers have the same molecular formula, but different structures. Conformers have different rotations around a single bond. The molecules are clearly not identical.
Example Question #222 : Gre Subject Test: Chemistry
Which of the following carbons represents the stereogenic center between the given isomers?


Carbon 1
Carbon 2
Carbon 3
Carbon 5
Carbon 4
Carbon 4
Epimers are isomers that have different configurations at only one carbon atom. This carbon atom is known as the stereogenic center. The given compounds are identical except for the orientation around carbon number 4; thus, carbon 4 is the stereogenic center.
Example Question #1 : Protein Structure
How many water molecules are lost from the condensation of 100 amino acids into a polypeptide?
A peptide bond is formed via the condensation of one amino acid's alpha-carboxy group with the alpha-amino group of another amino acid. Thus, the joining together of two amino acids results in the loss of one water molecule. Likewise, joining three amino acids together results in the loss of two water molecules. Following this pattern, we can conclude that the number of water molecules lost is equal to the number of amino acids joined together, minus 1. Therefore, the joining together of 100 amino acids results in the loss of 99 water molecules.
Example Question #202 : Organic Chemistry, Biochemistry, And Metabolism
An enzyme that cleaves disulfide bridges would most disrupt a protein containing which amino acid sequence?
Tyr–Cys–Cys–Thr–Val–Leu
Cys–Leu–Val–Tyr–Tyr–Thr
Val–Leu–Leu–Cys–Tyr–Thr
Tyr–Cys–Val–Val–Leu–Thr
All of the answers would be equally affected
Tyr–Cys–Cys–Thr–Val–Leu
Disulfide bridges are made between two cysteine amino acids. An enzyme that cleaves disulfide bonds would disrupt a protein containing the most cysteine residues; therefore, Tyr–Cys–Cys–Thr–Val–Leu is the correct answer.
Example Question #1 : Nucleic Acids
Of the following groups of nitrogenous bases, which does not contain a purine?
Thymine, adenine, cytosine
Cytosine, thymine, guanine
Cytosine, thymine, uracil
Adenine, guanine, thymine
Guanine, uracil, adenine
Cytosine, thymine, uracil
We can use the mnemonic "PurAG" to remember that the purines are adenine and guanine. The only choice that does not contain a purine, therefore, is "cytosine, thymine, and uracil." Remember, pyrimidines contain a single ring, while purines have two.
Example Question #52 : Organic Chemistry
Which of the following molecules is amphipathic?
Glutamate
Phospholipid
Triglyceride
Maltose
Phospholipid
Amphipathic molecules contain both polar and nonpolar regions, making them an extremely diverse class with an array of functions. For example, bile is an amphipathic molecule whose nonpolar region interacts with fats and whose polar region interacts with the aqueous environment of the small intestine.
Most lipids are entirely nonpolar and hydrophobic. Phospholipids, however, are formed from a glycerol molecule bound to two hydrophobic fatty acid tails and a hydrophilic phosphate head. This structure allows phospholipids amphipathic properties. Most notably, phospholipids are able to interact with the aqueous environments in the cell cytosol and extracellular environment, while maintaining the hydrophobic region of the cell membrane that acts as a semipermeable barrier.
Triglycerides are considered nonpolar. Glutamate is an acidic amino acid with highly polar properties. Maltose is a six-carbon sugar (carbohydrate) and is highly polar.
Example Question #1 : Lipids
Which of the following will you most likely find in a steroid molecule?
Nitrogenous base
Cyclohexane ring
Pentose sugar
Phosphate group
Cyclohexane ring
Steroids are a type of lipid that are characterized by their four-ring molecular structure. The four rings consist of three six-membered rings and one five-membered ring. Recall that six-membered rings are called cyclohexanes and five-membered rings are called cyclopentanes; therefore, you will most likely find a cyclohexane in a steroid.
Phosphate groups, pentose sugars, and nitrogenous bases are found in nucleotides, which are monomers that make up nucleic acids. Steroids are a type of lipid; therefore, you will most likely not find these substances in a steroid.
Example Question #6 : Carbohydrates
Which type of bond is created between carbohydrates and the sidechain amine of select asparagine residues in proteins?
N-glycosidic
Both are correct
Neither is correct
O-glycosidic
N-glycosidic
A glycosidic bond covalently joins a carbohydrate molecule to another molecule. An O-glycosidic bond is a covalent linkage between a carbohydrate and a protein, joining a serine or threonine hydroxyl side chain and a sugar (oxygen in the bond yields "O"). An N-glycosidic linkage involves bonding of a carbohydrate and a protein, joining an asparagine side chain amide and a sugar (nitrogen in the bond yields "N"). Thus, N-glycosidic is the correct answer.
Certified Tutor
Certified Tutor
All GRE Subject Test: Chemistry Resources