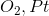All Organic Chemistry Resources
Example Questions
Example Question #1 : Using Pcc
What is the product of the reaction shown?
PCC can be used to oxidize primary alcohols into aldehydes, or secondary alcohols into ketones. The starting material shown is a secondary alcohol, so the product will be a ketone (a carbonyl (
Example Question #1 : Using Dichromate Compounds
A chemist adds the orange oxidizing agent, Na2Cr2O7, to the following substrates and dissolves the mixture in an aqueous solution of sulfuric acid. Oxidation is indicated by the disappearance of the orange color. Which of the substrate-oxidant solutions will remain orange?
All of the solutions will lose their orange color, indicating oxidation
I
IV
III
II
II
The following reaction schemes show the oxidation of all substrates, indicating that substrate II is in the highest oxidation state possible, and that an oxidation of this compound will not proceed.
Remember that in sulfuric acid and water, Na2Cr2O7 will be converted to CrO3, the active oxidant species. Furthermore, the oxidation mechanism involving this species includes the key step in which a hydrogen bonded to the carbon in question is eliminated, and simutaneously, a double bond from that carbon to an oxygen is installed. Thus, all substrates that feature at least one hydrogen bonded to the carbon to be oxidized can and will be oxidized in the precense of chromium trioxide.
Lastly, remember that these reactions are taking place in the prescence of water. While substrates such as compound III do not appear to be oxidizable, attack of water at the aldehyde carbon will give a dialcohol tetrathedral intermediate that can be immediately oxidized by chromium trioxide to the corresponding carboxylic acid. A similar mechanism occurs for substrate I, wherein, after the ketone oxidation state is achieved, an attack of water furnishes the same dialcohol intermediate that is oxidized to the carboxylic acid. Remember that the highest oxidation state available for organic compounds containing more than one carbon is the carboxylic acid oxidation state. Chromium trioxide will oxidize all organics to this oxidation state, unless directly-bonded hydrogens are not present in lower oxidation states, such as shown with substrate IV.

Example Question #12 : Organic Oxidizing Agents
Which of the following reagents would satisfy the given reaction?
In order to drive the reactant, we must first convert the alcohol group on the ethanol into a carboxylic acid. We do so by using the oxidizing agent, 

Example Question #11 : Organic Oxidizing Agents
Which of the following substrates will be oxidized into a ketone when reacting with 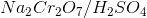
An aldehyde
A secondary alcohol
A primary alcohol
A carboxylic acid
A secondary alcohol

Not only can 
Example Question #2 : Using Dichromate Compounds

What would be the product of the given reaction?


![]()
![]()

The reaction given would give a ketone. This type of reaction is called an oxidation reaction. Oxidation of a secondary alcohol as in the reaction given by 

Example Question #1 : Using Other Organic Oxidizing Agents
Which of the following reagents can turn primary alcohols into a carboxylic acid?
PCC
Tollen's test
Jones Reagent (chromic acid in acetone)
Jones Reagent (chromic acid in acetone)
The Jones reagent can convert primary alcohol to acids and secondary alcohols to ketones. The Tollen's test only converts aldehydes to carboxylic acids. PCC can only convert primary and secondary alcohol to aldehydes and ketones, respectively. 

Example Question #2 : Using Other Organic Oxidizing Agents
What is an appropriate reagent to convert a primary alcohol to an aldehyde?
To form the aldehyde, the alcohol must be oxidized. However, potassium permanganate and chromic acid are too strong and would yield a carboxylic acid. Ozonolysis works with alkenes and oxygen over platinum would not react. PCC is correct because it will oxidize the alcohol to form an aldehyde but is too weak to continue on to form the carboxylic acid.
Example Question #1 : Using Sodium Borohydride
As a reducing agent, 
electron
proton
hydrogen molecule
hydrogen atom
hydride ion
hydride ion
Sodium borohydride donates a hydride ion to a ketone or aldehyde. In order to form a ketone or aldehyde, a nucleophile must attack the carbonyl group. This is because the ketone or aldehyde has an electrophilic carbon—a nucleophile must attack it in order for any reaction to occur. A hydride ion is the only answer choice that plays the role of a nucleophile.
Example Question #2 : Using Sodium Borohydride
Which of the following reaction conditions will selectively reduce the ketone in the following compound, retaining the alkene functionality?

Pd and H2 in hexanes
CeCl3 and NaBH4 in MeOH
LiAlH4 in THF
NaBH4 in MeOH
Pd, BaSO4, and H2 in hexanes
CeCl3 and NaBH4 in MeOH
The correct choice, CeCl3 and NaBH4 in MeOH, shows reagents know as "Luche conditions," which are able to modify the reactivity of sodium borohydride to reduce the carbonyl to an alcohol without affecting alkene groups. This occurs as the cerium ion coordinates strongly to the carbonyl oxygen, which subsequently greatly enhances the electrophilicity at the carbonyl carbon. Nucleophilic attack of the hydride readily occurs, simultaneously destroying the electropilicty of the beta carbon of the alkene, such that it will not be reduced by the hydride reagent.
The incorrect answer choices would give various undesired products as detailed below:
NaBH4 in MeOH
Use of unmodified sodium borohydride would result in a 1,4 conjugate addition reaction, saturating the alkene, with a subsequent reduction of the ketone to an alcohol.
LiAlH4 in THF
Use of lithium aluminum hydride would give the same product as use of unmodified sodium borohydride, following the same reduction mechanism.
Pd and H2 in hexanes
This reagent will give reduction of the alkene only.
Pd, BaSO4, and H2 in hexanes
This reagent combination, known as Lindlar's catalyst, will also reduce the alkene only. This reagent is typically used to selectively reduce an alkyne to an alkene.
Example Question #462 : Organic Chemistry
Which of these can be reduced by sodium borohydride?
3-pentanone
2-butene
Propanoic acid
2-butanol
None of these
3-pentanone
Sodium borohydride is a reducing agent with formula 
Certified Tutor
All Organic Chemistry Resources