All Physical Chemistry Resources
Example Questions
Example Question #2 : Orbitals And Hybridization
What is true when comparing the electron configuration of elemental sodium 

Elemental sodium is paramagnetic
The sodium ion has one additional electron in an 
The outermost shell of the sodium ion has one electron
The sodium ion has more electrons in 
Elemental sodium is paramagnetic
To answer this question, we need to find the electron configuration of both elemental sodium and sodium cation. If we look at the periodic table we can see that sodium is found on the first column. Since it is found in the first column, sodium has one valence electron. To complete octet, sodium will readily lose an electron and become a positively charged sodium ion. The electron configuration for sodium is 






Example Question #11 : Atoms And Elements
It is observed that a molecule has three hybridized orbitals in its outermost shell. What can you conclude about this molecule?
It has a lone pair electron
It has four single bonds
It has a double bond
None of these
It has a double bond
Hybridization is a process involving the fusion, or hybridization, of 














































Example Question #3 : Orbitals And Hybridization
Which of the following is true regarding carbon tetrachloride?
Hybridization in this molecule involves three times as many 

The carbon in this molecule has similar hybridization as the carbon in carbon dioxide
Hybridization in this molecule involves two times as many 

More than one of these are true
Hybridization in this molecule involves three times as many 

Carbon tetrachloride, 














Example Question #1 : Valence Electrons
How many valence electrons are in an atom of phosphorus?
Thirteen
Five
Three
Eight
Five
When determining the number of valence electrons for an atom, simply count the number of electrons present in the outermost shell's s and p orbitals. Phosphorus has two electrons in the 3s subshell, and three more in the 3p subshell, making a total of five valence electrons.
Example Question #101 : Physical Chemistry
What is the complete electron configuration for the sulfur atom?
1s22s22p63s23p2
1s22s22p63s23p4
1s22s22p63s23p2
1s42p63s23p4
1s42p63s23p4
1s22s22p63s23p4
The types of subshells, from smallest to largest, are as follows: s, p, d, and f. These four subshells correspond respectively to the following quantum numbers: 0, 1, 2, and 3. From the periodic table, it is known that sulfur has 16 electrons. Additionally, the maximum number of electrons the s sublevel can hold is 2. The maximum number of electrons that the p subshell can hold is 6, and electrons fill orbitals from lowest to highest energy. 1s22s22p63s23p4 is the only choice that meets the criteria.
Example Question #1 : Valence Electrons
Arrange the following ions in order of decreasing ionic radius: nitride ion, oxide ion, sodium ion, aluminum ion.
The ions have the same ionic radius.
Notice that all the ions have the same electron configuration as neon: 1s22s22p6. Substances with the same number of electrons and the same electron configuration are isoelectronic, meaning the number of electrons is the same but the number of protons is not. In an isoelectronic series, the ion with the most protons is smallest because the nucleus exerts a stronger force of attraction and the electrons are pulled closer to the nucleus. Consequently, the ion with the fewest protons is largest. 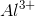
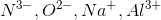
Example Question #11 : Atoms And Elements
Which of the following trends decreases as you move from left to right on the periodic table?
Atomic radius
Electron affinity
Electronegativity
Ionization energy
Atomic radius
Although it may seem counterintuitive, atomic radius does decrease from left to right on the periodic table. The reason for this is because the added positive charge in the nucleus causes the elctrons to be pulled more strongly towards the center, which decreases the atomic radius.
Example Question #111 : Physical Chemistry
Which of the following atoms has the greatest electronegativity?
Arsenic
Selenium
Sulfur
Phosphorus
Sulfur
Electronegativity values become greater as you move up and to the right on the periodic table. Of the four atoms listed, sulfur is the highest up and farthest to the right, giving it the greatest electronegativity.
Example Question #12 : Atoms And Elements
Which of these is in order of increasing ionization energy?
Ionization energy is the amount of energy that an atom in the ground state must absorb to emit an electron. Upon ionization, a cation is formed. Ionization energy increases from bottom to top within a group, and from left to right within a row of the periodic table which is the opposite trend that atomic radius follows. Referring to the periodic table, we can see that of these group VII elements, fluorine has the highest ionization energy.
Example Question #13 : Atoms And Elements
Which of the following lists is arranged in order of increasing atomic radius?
All of these atoms are in group VII. Within a group, atomic radii increase from top to bottom due to the increased number of electron shells.
Certified Tutor
All Physical Chemistry Resources


















