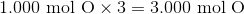Empirical and Molecular Formulas - AP Chemistry
Card 0 of 20
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is  , which can be verified by dividing the coefficients of the molecular formula by 6. Note that
, which can be verified by dividing the coefficients of the molecular formula by 6. Note that 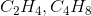 is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula
is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula  .
.
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is 
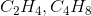

Compare your answer with the correct one above
Vanillin is composed of  carbon,
carbon,  oxygen, and
oxygen, and  hydrogen.
hydrogen.
What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:

Vanillin is composed of 


What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is  carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:
carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:



After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:



We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.


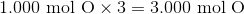
This gives us  as the final answer.
as the final answer.
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is 
After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:
We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.
This gives us 
Compare your answer with the correct one above
What is the correct molecular formula for phosphoric acid?
What is the correct molecular formula for phosphoric acid?
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is  .
.  is the molecular formula for hydrophosphoric acid,
is the molecular formula for hydrophosphoric acid,  is the molecular formula for phosphorous acid, and
is the molecular formula for phosphorous acid, and  is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is 



Compare your answer with the correct one above
A molecule with the empirical formula  has a molar mass of
has a molar mass of  . Find its molecular formula.
. Find its molecular formula.
A molecule with the empirical formula 

Start by finding the molar mass of of  by adding up the molar masses of its constituent atoms.
by adding up the molar masses of its constituent atoms.

Now, divide the molar mass of the molecule by the molar mass of its empirical formula.

In order to find the molecular formula, you will need to multiply the empirical formula by  . Thus, the molecular formula is
. Thus, the molecular formula is  .
.
Start by finding the molar mass of of 
Now, divide the molar mass of the molecule by the molar mass of its empirical formula.
In order to find the molecular formula, you will need to multiply the empirical formula by 

Compare your answer with the correct one above
What is the empirical formula for a compound that contains  carbon and
carbon and  hydrogen?
hydrogen?
What is the empirical formula for a compound that contains 

We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with  of the starting material. That way, we can convert each percentage directly into grams.
of the starting material. That way, we can convert each percentage directly into grams.
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.


In other words, what we have found is that in our compound, for every  of carbon, there will also be
of carbon, there will also be  of hydrogen.
of hydrogen.

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.

What this tells us is that for every  of carbon in this compound, there will be
of carbon in this compound, there will be  of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with 
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.
In other words, what we have found is that in our compound, for every 

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.
What this tells us is that for every 

Compare your answer with the correct one above
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is  , which can be verified by dividing the coefficients of the molecular formula by 6. Note that
, which can be verified by dividing the coefficients of the molecular formula by 6. Note that 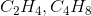 is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula
is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula  .
.
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is 
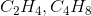

Compare your answer with the correct one above
Vanillin is composed of  carbon,
carbon,  oxygen, and
oxygen, and  hydrogen.
hydrogen.
What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:

Vanillin is composed of 


What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is  carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:
carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:



After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:



We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.


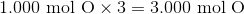
This gives us  as the final answer.
as the final answer.
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is 
After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:
We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.
This gives us 
Compare your answer with the correct one above
What is the correct molecular formula for phosphoric acid?
What is the correct molecular formula for phosphoric acid?
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is  .
.  is the molecular formula for hydrophosphoric acid,
is the molecular formula for hydrophosphoric acid,  is the molecular formula for phosphorous acid, and
is the molecular formula for phosphorous acid, and  is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is 



Compare your answer with the correct one above
A molecule with the empirical formula  has a molar mass of
has a molar mass of  . Find its molecular formula.
. Find its molecular formula.
A molecule with the empirical formula 

Start by finding the molar mass of of  by adding up the molar masses of its constituent atoms.
by adding up the molar masses of its constituent atoms.

Now, divide the molar mass of the molecule by the molar mass of its empirical formula.

In order to find the molecular formula, you will need to multiply the empirical formula by  . Thus, the molecular formula is
. Thus, the molecular formula is  .
.
Start by finding the molar mass of of 
Now, divide the molar mass of the molecule by the molar mass of its empirical formula.
In order to find the molecular formula, you will need to multiply the empirical formula by 

Compare your answer with the correct one above
What is the empirical formula for a compound that contains  carbon and
carbon and  hydrogen?
hydrogen?
What is the empirical formula for a compound that contains 

We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with  of the starting material. That way, we can convert each percentage directly into grams.
of the starting material. That way, we can convert each percentage directly into grams.
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.


In other words, what we have found is that in our compound, for every  of carbon, there will also be
of carbon, there will also be  of hydrogen.
of hydrogen.

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.

What this tells us is that for every  of carbon in this compound, there will be
of carbon in this compound, there will be  of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with 
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.
In other words, what we have found is that in our compound, for every 

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.
What this tells us is that for every 

Compare your answer with the correct one above
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is  , which can be verified by dividing the coefficients of the molecular formula by 6. Note that
, which can be verified by dividing the coefficients of the molecular formula by 6. Note that 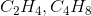 is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula
is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula  .
.
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is 
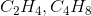

Compare your answer with the correct one above
Vanillin is composed of  carbon,
carbon,  oxygen, and
oxygen, and  hydrogen.
hydrogen.
What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:

Vanillin is composed of 


What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is  carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:
carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:



After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:



We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.


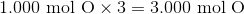
This gives us  as the final answer.
as the final answer.
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is 
After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:
We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.
This gives us 
Compare your answer with the correct one above
What is the correct molecular formula for phosphoric acid?
What is the correct molecular formula for phosphoric acid?
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is  .
.  is the molecular formula for hydrophosphoric acid,
is the molecular formula for hydrophosphoric acid,  is the molecular formula for phosphorous acid, and
is the molecular formula for phosphorous acid, and  is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is 



Compare your answer with the correct one above
A molecule with the empirical formula  has a molar mass of
has a molar mass of  . Find its molecular formula.
. Find its molecular formula.
A molecule with the empirical formula 

Start by finding the molar mass of of  by adding up the molar masses of its constituent atoms.
by adding up the molar masses of its constituent atoms.

Now, divide the molar mass of the molecule by the molar mass of its empirical formula.

In order to find the molecular formula, you will need to multiply the empirical formula by  . Thus, the molecular formula is
. Thus, the molecular formula is  .
.
Start by finding the molar mass of of 
Now, divide the molar mass of the molecule by the molar mass of its empirical formula.
In order to find the molecular formula, you will need to multiply the empirical formula by 

Compare your answer with the correct one above
What is the empirical formula for a compound that contains  carbon and
carbon and  hydrogen?
hydrogen?
What is the empirical formula for a compound that contains 

We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with  of the starting material. That way, we can convert each percentage directly into grams.
of the starting material. That way, we can convert each percentage directly into grams.
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.


In other words, what we have found is that in our compound, for every  of carbon, there will also be
of carbon, there will also be  of hydrogen.
of hydrogen.

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.

What this tells us is that for every  of carbon in this compound, there will be
of carbon in this compound, there will be  of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with 
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.
In other words, what we have found is that in our compound, for every 

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.
What this tells us is that for every 

Compare your answer with the correct one above
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
Which pair of formulas represents the empirical formula and molecular formula for a certain compound?
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is  , which can be verified by dividing the coefficients of the molecular formula by 6. Note that
, which can be verified by dividing the coefficients of the molecular formula by 6. Note that 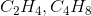 is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula
is not correct because neither of the formulas is an empirical formula; they are both possible molecular formulas for a compound with the empirical formula  .
.
To find an empirical formula, take a molecular formula and divide the subscript of each element by the greatest common factor of all the subscripts. In this case, the only pair that works is 
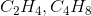

Compare your answer with the correct one above
Vanillin is composed of  carbon,
carbon,  oxygen, and
oxygen, and  hydrogen.
hydrogen.
What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:

Vanillin is composed of 


What is the empirical formula of vanillin?
Refer to the following table for the atomic masses of the elements shown:
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is  carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:
carbon; therefore, the estimated sample would contain 66.3 g of carbon. After doing this for each of the elements vanillin contains, we can use the atomic mass of carbon to find how many moles of carbon make up the sample. We then do the same for each element that composes vanillin. See calculations below:



After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:



We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.


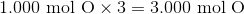
This gives us  as the final answer.
as the final answer.
In order to calculate the empirical formula of vanillin from the given percent composition data, we can pretend we have a 100 g sample vanillin and from there estimate how many grams of each element make up the sample. For example, Vanillin is 
After finding the number of moles of each element in the hypothetical 100 g sample, we look at the ratio between the moles of each element by dividing all the samples by smallest number of moles. After finding the ratios of the samples, we are able to use the empirical formula of the vanillin by rounding up to the nearest whole number for each sample. If the ratios are not close enough to round up (like in this example), we multiply the ratios all by the same number until each number is equal to a whole number nicely. See calculations below:
We need whole numbers, so multiply each of the resulting numbers by 3 to get rid of the fractions.
This gives us 
Compare your answer with the correct one above
What is the correct molecular formula for phosphoric acid?
What is the correct molecular formula for phosphoric acid?
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is  .
.  is the molecular formula for hydrophosphoric acid,
is the molecular formula for hydrophosphoric acid,  is the molecular formula for phosphorous acid, and
is the molecular formula for phosphorous acid, and  is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
is just a charged phosphate ion, which has biochemical significance with regards to DNA structure and energy molecules like ATP. Phosphorus is not a diatomic atom.
Phosphoric acid combines a single phosphate ion with three hydrogen ions. The correct molecular formula is 



Compare your answer with the correct one above
A molecule with the empirical formula  has a molar mass of
has a molar mass of  . Find its molecular formula.
. Find its molecular formula.
A molecule with the empirical formula 

Start by finding the molar mass of of  by adding up the molar masses of its constituent atoms.
by adding up the molar masses of its constituent atoms.

Now, divide the molar mass of the molecule by the molar mass of its empirical formula.

In order to find the molecular formula, you will need to multiply the empirical formula by  . Thus, the molecular formula is
. Thus, the molecular formula is  .
.
Start by finding the molar mass of of 
Now, divide the molar mass of the molecule by the molar mass of its empirical formula.
In order to find the molecular formula, you will need to multiply the empirical formula by 

Compare your answer with the correct one above
What is the empirical formula for a compound that contains  carbon and
carbon and  hydrogen?
hydrogen?
What is the empirical formula for a compound that contains 

We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with  of the starting material. That way, we can convert each percentage directly into grams.
of the starting material. That way, we can convert each percentage directly into grams.
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.


In other words, what we have found is that in our compound, for every  of carbon, there will also be
of carbon, there will also be  of hydrogen.
of hydrogen.

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.

What this tells us is that for every  of carbon in this compound, there will be
of carbon in this compound, there will be  of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
of hydrogen as well. This is equivalent to the previous ratio, but now it has been reduced to the lowest possible whole numbers.
We're given the percent composition of the elements carbon and hydrogen in a given compound, and we're asked to determine the empirical formula.
The first thing we have to do is determine the relative molar ratios of each element. In this case, because we are given the percent composition, we can assume that we're starting off with 
Following that, we need to use the molar mass of each element in order to convert it from grams to moles.
In other words, what we have found is that in our compound, for every 

But remember, the empirical formula for any given compound tells us the lowest whole number ratio of the compound's constituent elements. Therefore, we'll need to divide each value by the lowest number out of all the values available.
What this tells us is that for every 

Compare your answer with the correct one above