0%
0 / 15 answered
Drawing Conclusions & Evaluating Claims Practice Test
•15 QuestionsQuestion
1 / 15
Q1
PASSAGE I
CHEMISTRY: This passage is adapted from a study on the solubility of various substances in water.
Solubility is defined as the maximum amount of a solute (substance being dissolved) that can dissolve in a specific amount of solvent (usually water) at a given temperature. The solubility of most solids increases with temperature, while the solubility of most gases decreases with temperature. A student performed an experiment to measure the solubility of three solid salts—Potassium Nitrate ($KNO_3$), Sodium Chloride ($NaCl$), and Cerium(III) Sulfate ($Ce_2(SO_4)_3$)—and one gas, Oxygen ($O_2$).
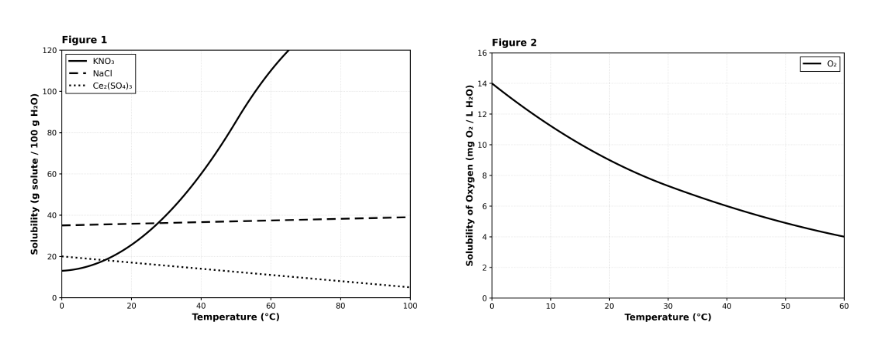
A student dissolves 50 g of KNO₃ in 100 g of water at 40°C. Based on Figure 1, the resulting solution would be best described as:
A student dissolves 50 g of KNO₃ in 100 g of water at 40°C. Based on Figure 1, the resulting solution would be best described as: