Variables & Controls
Help Questions
ACT Science › Variables & Controls
Which of the following variables was intentionally held constant in the experiment that produced the data in Table 1?
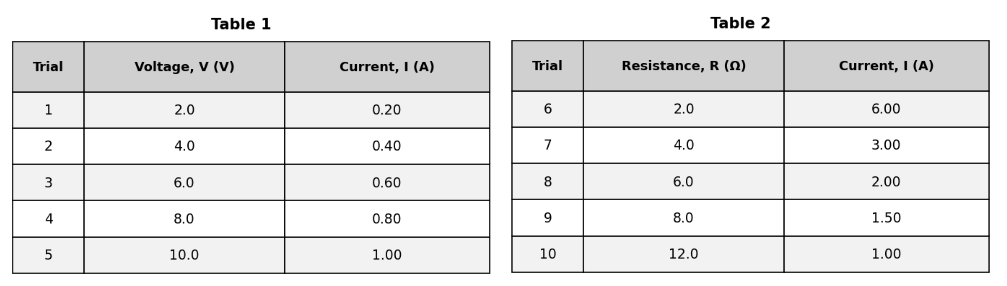
The type of power supply
The current (I)
The voltage (V)
The resistance (R)
Explanation
The correct answer is C (the resistance R). The introduction to Table 1 explicitly states: 'the student used a resistor with a constant resistance of 10.0 Ω.' This was held fixed so that the effect of changing voltage on current could be isolated. A (current) is the dependent variable being measured — it changes in response to voltage changes and is not held constant. B (voltage) is the independent variable being deliberately varied across trials. H correctly identifies resistance as the controlled constant. D (type of power supply) is not mentioned as a variable in the study and while presumably constant, it is not the primary experimental control that was intentionally fixed to isolate the relationship. On controlled-variable questions, look for the explicit statement in the study introduction identifying what was kept constant.
During gel electrophoresis, which property of the DNA fragments primarily determines how far they migrate through the gel?
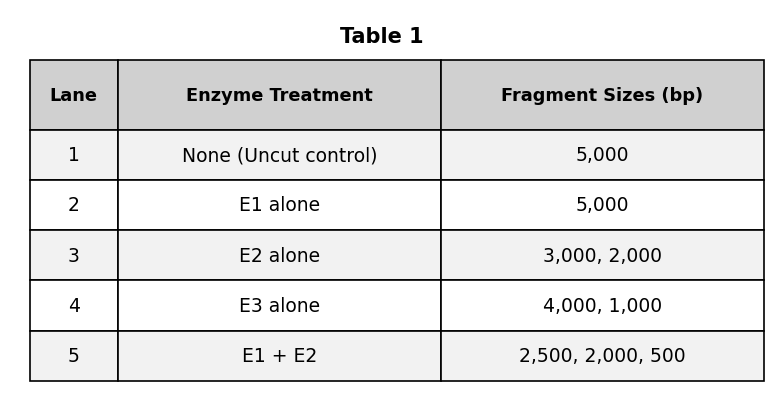
Their rate of mutation
Their restriction enzyme binding site
Their electrical charge
Their length in base pairs
Explanation
The correct answer is C (their length in base pairs). The passage explicitly states: 'Smaller DNA fragments move through the gel much faster and travel further than larger fragments.' Size — measured in base pairs — is the primary determinant of migration distance. All DNA fragments carry a negative charge (this is why they migrate toward the positive electrode), so electrical charge is essentially the same for all fragments and does not distinguish how far they travel. F (electrical charge) is what causes all DNA to move toward the positive electrode, but it does not explain why different-sized fragments travel different distances. G (restriction enzyme binding sites) are the sequences where enzymes cut the DNA — these are properties of the sequence, not migration distance. J (rate of mutation) is irrelevant to electrophoresis migration.
A microbiology student tested whether disinfectant concentration affects bacterial survival on plastic. She prepared four spray bottles containing the same disinfectant at 0%, 25%, 50%, and 75% concentration (diluted with sterile water). Identical plastic squares were inoculated with the same volume of bacterial culture and left for 5 minutes. Each square was sprayed with exactly 2.0 mL of solution from its bottle and left for 2 minutes before being swabbed. The swab was streaked onto agar plates that were incubated at 37°C for 24 hours. The student counted the number of colonies that grew on each plate. In this experiment, the dependent variable is:
Disinfectant concentration used
Volume sprayed onto each plastic square
Incubation temperature (37°C)
Number of bacterial colonies after incubation
Explanation
In this experiment, the independent variable is the disinfectant concentration (0%, 25%, 50%, 75%), the dependent variable is the number of bacterial colonies after incubation, and controlled variables include the volume sprayed (2.0 mL), incubation temperature (37°C), plastic square type, bacterial culture volume, inoculation time (5 minutes), exposure time (2 minutes), agar plates, and incubation time (24 hours). The independent variable is manipulated to test its efficacy, the dependent variable is counted to assess bacterial survival, and controlled variables are maintained constant to ensure the results reflect only the concentration's impact. Choice B correctly identifies the number of bacterial colonies as the dependent variable because it was the measured outcome responding to different concentrations. Choice D is a distractor as it represents a controlled variable, the fixed incubation temperature, not the dependent variable.
A student investigated how salt concentration affects the boiling point of water. She prepared four 250 mL beakers, each containing 200 mL of distilled water. She added table salt to make solutions of 0%, 2%, 4%, and 6% salt by mass. Each beaker was heated on the same hot plate setting, starting at room temperature (22°C). The student stirred each solution at a constant rate with the same glass rod and recorded the temperature when the liquid reached a steady rolling boil for 30 seconds. She used the same thermometer for all trials and measured boiling point to the nearest 0.1°C. Which of the following is the independent variable?
Hot plate model used
Volume of water in each beaker
Temperature at which boiling began
Salt concentration of the solution
Explanation
In this experiment, the independent variable is the salt concentration of the solution, which was varied at 0%, 2%, 4%, and 6%; the dependent variable is the temperature at which boiling began, which was measured for each solution; and controlled variables include the volume of water (200 mL in each beaker), the hot plate model and setting, the starting temperature (22°C), stirring rate, and thermometer used. The independent variable is the factor deliberately manipulated by the experimenter to observe its effect, the dependent variable is the outcome measured to see the response, and controlled variables are kept constant across trials to ensure the results are due to the independent variable alone. Choice C correctly identifies the salt concentration as the independent variable because it was systematically changed while other factors were held steady. Choice D is a distractor as it represents the dependent variable, which is the measured boiling temperature, not the manipulated factor.
A student tested how the temperature of water affects the time it takes a tablet to dissolve. She placed one identical effervescent tablet into 200 mL of water at each of four temperatures: 10°C, 20°C, 30°C, and 40°C. She used the same beaker type, did not stir the water in any trial, and used the same brand of tablets. She started a timer when the tablet touched the water and stopped the timer when bubbling ceased and no solid remained. Which of the following is the independent variable?
Water temperature
Time for the tablet to dissolve
Volume of water (200 mL)
Brand of effervescent tablet
Explanation
In this experiment, the independent variable is the water temperature (10°C, 20°C, 30°C, and 40°C), the dependent variable is the time for the tablet to dissolve, and controlled variables include the volume of water (200 mL), beaker type, lack of stirring, tablet brand, and tablet size (identical effervescent tablets). The independent variable is the factor varied to test its impact, the dependent variable is the measured effect of that variation, and controlled variables are maintained constant to ensure the experiment's validity. Choice B correctly identifies water temperature as the independent variable because it was systematically changed to observe its effect on dissolution time. Choice C is a distractor as it represents a controlled variable, the fixed water volume, which was not manipulated.
A student investigated how fertilizer amount affects algae growth in small aquariums. She set up four identical 2.0 L tanks, each filled with 1.5 L of the same pond water and the same initial algae sample. She added fertilizer at 0 g, 0.5 g, 1.0 g, or 1.5 g per tank. All tanks were kept under the same lamp, at the same distance from the light, and at 24°C. After 7 days, she measured algae growth by filtering each tank and recording the dry mass of algae (g). The experimenter manipulated which factor and measured which factor?
Manipulated algae dry mass; measured fertilizer amount
Manipulated temperature; measured light distance
Manipulated fertilizer amount; measured algae dry mass
Manipulated pond water volume; measured tank size
Explanation
In this experiment, the independent variable is the fertilizer amount (0 g, 0.5 g, 1.0 g, or 1.5 g), the dependent variable is the algae dry mass after 7 days, and controlled variables include the tank size (2.0 L), pond water volume (1.5 L), initial algae sample, temperature (24°C), light distance, and lamp used. The independent variable is manipulated to observe its effects, the dependent variable is measured to quantify the response, and controlled variables are held constant to ensure the results stem solely from the manipulation. Choice B correctly identifies that the experimenter manipulated the fertilizer amount and measured the algae dry mass, matching the experimental design. Choice D is a key distractor as it reverses the variables, incorrectly stating algae mass was manipulated and fertilizer measured.
A chemist studied how stirring speed affects the time required for 5.0 g of sugar to dissolve in 100 mL of water. She used a magnetic stir plate and tested four stirring speeds: 0 rpm, 200 rpm, 400 rpm, and 600 rpm. The water temperature was kept constant at 25°C using a water bath, and the same beaker type and sugar brand were used each time. For each trial, the chemist started a stopwatch when sugar was added and stopped it when no visible crystals remained. Which of the following is the independent variable?
Water temperature (25°C)
Mass of sugar added
Time for the sugar to dissolve
Stirring speed (rpm)
Explanation
In this experiment, the independent variable is the stirring speed (0 rpm, 200 rpm, 400 rpm, and 600 rpm), the dependent variable is the time for the sugar to dissolve, and controlled variables include the mass of sugar (5.0 g), water volume (100 mL), water temperature (25°C), beaker type, and sugar brand. The independent variable is the factor the experimenter changes on purpose, the dependent variable is what is observed and recorded as the result, and controlled variables are maintained the same in all trials to prevent them from affecting the outcome. Choice C correctly identifies stirring speed as the independent variable because it was deliberately varied to study its effect on dissolution time. Choice A is a distractor as it represents a controlled variable, the water temperature kept at 25°C, not the manipulated factor.
A food science student tested how baking temperature affects cookie spread. She prepared one batch of dough and used a scoop to place identical 30 g dough balls on the same type of baking sheet lined with parchment. She baked trays at 160°C, 180°C, 200°C, and 220°C for exactly 10 minutes in the same oven, allowing the oven to preheat fully each time. After cooling for 5 minutes, she measured cookie diameter (cm) with a ruler. Which variable was held constant?
Amount of cookie spread
Cookie diameter after cooling
Baking temperature
Mass of each dough ball (30 g)
Explanation
In this experiment, the independent variable is the baking temperature (160°C, 180°C, 200°C, 220°C), the dependent variable is the cookie diameter after cooling (or amount of spread), and controlled variables include the mass of each dough ball (30 g), baking time (10 minutes), dough batch, baking sheet type, parchment lining, oven, and cooling time (5 minutes). The independent variable is altered by the experimenter, the dependent variable is measured to see the resulting change, and controlled variables are kept the same across trials to attribute differences solely to the independent variable. Choice C correctly identifies the mass of each dough ball as a variable held constant because it was fixed at 30 g for all cookies to isolate temperature's effect. Choice D is a distractor as it represents the dependent variable, the measured diameter, which varied rather than being controlled.
A chemistry student examined whether stirring rate changes how quickly sugar dissolves. She added 10.0 g of granulated sugar to 200 mL of water at $25^\circ\text{C}$ in identical beakers. Using the same magnetic stirrer, she tested four stirring settings: 0 rpm (no stirring), 200 rpm, 400 rpm, and 600 rpm. She recorded the time (in seconds) until no visible sugar crystals remained. The sugar mass, water volume, water temperature, beaker type, and observation method were kept the same each trial. The experimenter manipulated and measured .
time to dissolve; sugar mass
water temperature; stirring rate
beaker type; water volume
stirring rate; time to dissolve
Explanation
In this experiment, the independent variable is the stirring rate, the dependent variable is the time to dissolve, and controlled variables include water temperature, sugar mass, water volume, and beaker type. The independent variable is deliberately varied by the experimenter, the dependent variable is the response that is measured, and controlled variables are maintained constant to ensure the test focuses only on the independent variable's effect. Choice B correctly identifies that the experimenter manipulated the stirring rate (0, 200, 400, 600 rpm) and measured the time to dissolve, matching the roles of independent and dependent variables. Choice A incorrectly swaps the roles, as water temperature was controlled, not manipulated.
A student tested the hypothesis that increasing salt concentration lowers the freezing point of water. She prepared four 100 mL samples of distilled water in identical cups and added NaCl to make solutions of 0%, 2%, 4%, and 6% (by mass). Each cup contained a thermometer, and all cups were placed in the same freezer set to $-15^\circ\text{C}$. The student stirred each sample for 10 s every minute and recorded the temperature at which the first ice crystals appeared (freezing point). The cup type, water volume, freezer setting, and stirring schedule were kept the same for all trials. Which of the following is the independent variable?
Salt concentration in the water
Freezer temperature setting
Temperature when ice first appears
Time spent stirring each minute
Explanation
In this experiment, the independent variable is the salt concentration in the water, the dependent variable is the temperature when ice first appears, and controlled variables include the freezer temperature setting, water volume, cup type, and stirring schedule. The independent variable is what the experimenter deliberately changes to test its effect, the dependent variable is the outcome measured in response to that change, and controlled variables are factors kept constant to ensure a fair test. Choice C correctly identifies the salt concentration as the independent variable because it was manipulated across different levels (0%, 2%, 4%, 6%) while observing the freezing point. Choice B represents the dependent variable, as it was the measured response, not what was changed.